FibroSIGHT™ Plus
Test Introduction
FibroSIGHT™ Plus leverages our stain-free Second Harmonic Generation (SHG) and Two-Photon Excitation Fluorescence (TPEF) imaging technology to capture detailed tissue structure and collagen fibers with high consistency – eliminating the variability associated with conventional tissue staining.
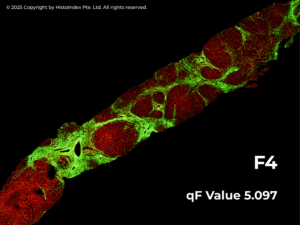
At the core of FibroSIGHT Plus is qFibrosis®, our proprietary AI-based algorithm, which automatically identifies and quantifies multiple fibrosis-related collagen parameters from SHG images. These are integrated into a continuous numerical index known as the qFibrosis® value, offering a detailed and objective measurement of fibrosis severity across the wide spectrum of fibrosis in MASH.
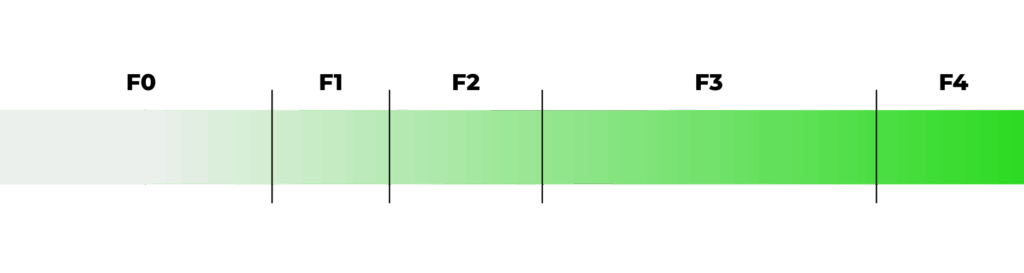
To support clinical decision-making, the qFibrosis value is further categorized into qualitative stages that align with the NASH-CRN fibrosis scoring system, providing both clinical relevance and improved resolution for tracking disease progression or treatment response.
How FibroSIGHT™ Plus Works
Capture Fibrosis with Advanced IMAGING Technology
FibroSIGHT Plus begins with a dual-channel imaging approach using Second Harmonic Generation (SHG) and Two-Photon Excitation Fluorescence (TPEF). These high-resolution, label-free techniques visualize liver biopsy samples without staining. TPEF captures anatomical structures like portal tracts (PT) and central veins (CV), enabling precise tissue zoning. The highly specific SHG channel provides accurate identification of collagen fibers, enabling identification and quantification of morphological and spatial metrics of fibrosis parameters.

QUANTIFYING Fibrosis with a Continuous qFibrosis value
Leveraging SHG images the qFibrosis algorithm automatically quantifies multiple collagen parameters and translates them into a continuous numerical index – the qFibrosis® value. This value captures both the severity and spatial distribution of fibrosis across the entire tissue sample. By offering greater granularity than traditional categorical scores, qFibrosis value enables clinicians to better characterize individual patients at diagnosis and to monitor subtle changes in their disease over time – such as early progression or regression – that may be missed with conventional methods.

TRANSLATING Scores into Clinically Interpretable Stages
The qFibrosis value is translated to ordinal stages that correspond to the NASH-CRN fibrosis stages to offer clinical interpretability. Through this multi-step, quantitative approach, FibroSIGHT Plus provides a reproducible and objective fibrosis assessment that enhances diagnostic accuracy and supports therapeutic decision making.
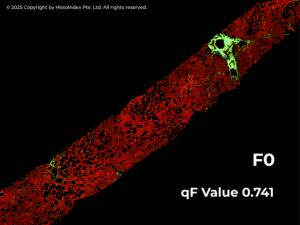

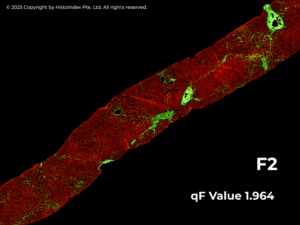
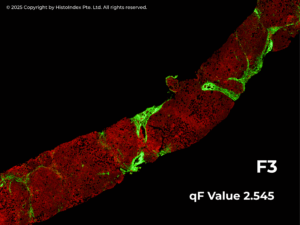
Structured REPORTING for Confident Decision-Making
FibroSIGHT™ Plus leverages our advanced proprietary stain-free imaging and digital pathology platform developed on Second Harmonic Generation (SHG) and Two-Photon Excitation Fluorescence (TPEF). SHG imaging detects fibrillar collagen in biological tissues with consistency, high-resolution and excellent signal-to-noise ratio, while TPEF allows visualization of pertinent cell structures – all without the use of any exogenous dyes. This overcomes the limitations and variability of traditional staining methods.
How FibroSIGHT™ Plus Performs in Real-World Clinical Cohorts
The qFibrosis algorithm has been extensively validated, showing strong correlation with the NASH-CRN staging system and reinforcing its clinical relevance6.
For detecting at-risk fibrosis (F2–F4) vs. early-stage (F0/1), it achieved an AUROC of 0.88, with 85% sensitivity and 71% specificity. For identifying cirrhosis (F4), it reached an AUROC of 0.91 with 96% specificity, enabling confident diagnosis of advanced disease.
These results confirm qFibrosis as a reliable and accurate tool for fibrosis stratification in clinical and research settings.
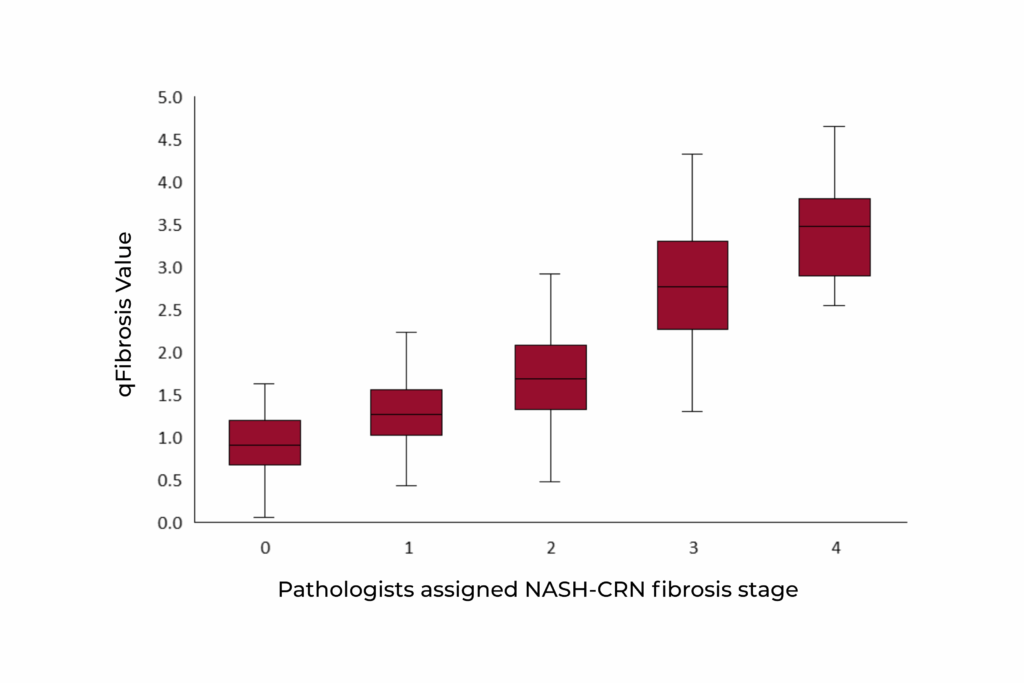
Correlation of qFibrosis with pathologist staging with NASH-CRN staging system (rs = 0.81, p-value < 0.001)
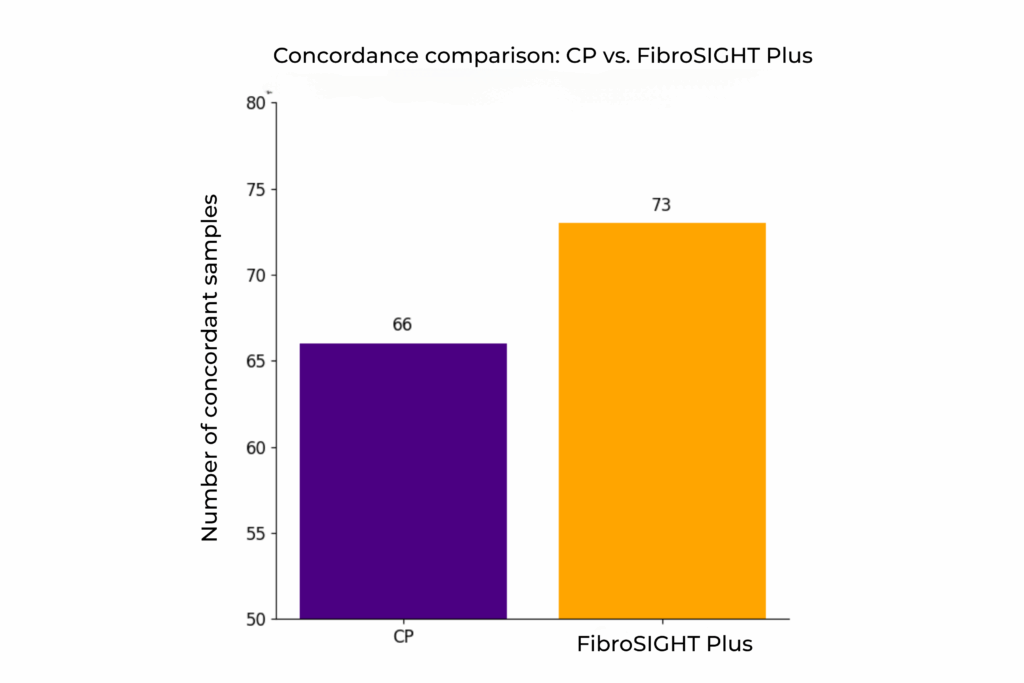
Concordance of community pathologists (CP) and FibroSIGHT Plus to the reference fibrosis read.
An internal validation study of 120 MASH samples demonstrated that fully automated fibrosis assessment with FibroSIGHT Plus yielded approximately 10% more samples with concordance to reference fibrosis stages – defined by a panel of expert pathologists – compared to assessments by community pathologists using traditional Masson’s Trichrome staining, which represents the current standard of care.
This underscores the qFibrosis algorithm’s utility as a more consistent and reliable tool for fibrosis staging today.
Sample Collection and Shipping Instructions
References
Test Ordering
The test may be ordered through the HistoIndex Website.This test was developed, and its analytical performance characteristics have been determined by PacificDx. It has not been cleared or approved by FDA. This assay has been validated pursuant to the CLIA regulations and is used for clinical purposes.

